 Problem P. 4100. (October 2008)
Problem P. 4100. (October 2008)
P. 4100. A wall divides the cylindrical, thermally insulated container of volume 20 litres into two parts as shown in the figure. There is vacuum in the left part which is closed by a thermally insulated fixed piston, and in the other part there is Nitrogen gas at a pressure of p=5.105 and at a temperature of 300 K. (The external pressure is 105 Pa.) The door on the wall opens because of the excess pressure, and the gas expands immediately. Then the fixing of the piston is ceased, and the gas is pushed back into its original place.
a) How much work was performed during the compression of the gas?
b) What are the temperature and the pressure of the gas at the end of the process?
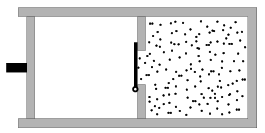
(5 pont)
Deadline expired on November 11, 2008.
Statistics:
106 students sent a solution. 5 points: Bali-Papp Donát, Berecz Dénes, Börcsök Bence, Csopor Dávid, Dinh Hoangthanh Attila, Földes Imre, Galzó Ákos Ferenc, Hartstein Máté, Jéhn Zoltán, Kótai Áron, Kovács Benjámin, Krämer Zsolt, Laczkó Zoltán Balázs, Lovas Lia Izabella, Márkus Bence Gábor, Németh Gábor , Patartics Bálint, Pázmán Koppány, Tamási Mátyás, Varga 777 Ádám, Varju 105 Tamás. 4 points: Balogh Gábor, Batki Bálint, Bodosi Eszter, Filep Gábor, Filep Tibor, Klenk 191 Blanka, Kunsági-Máté Éva, Maknics András, Mayer Martin János, Meláth Kamilla, Müller Márk, Nagy 668 Balázs, Najbauer Eszter Éva, Pap Máté, Para Attila, Szatmári András Zoltán, Török 999 Csaba, Vuchetich Bálint, Wang Daqian, Zsámboki Richárd. 3 points: 1 student. 2 points: 8 students. 1 point: 5 students. 0 point: 51 students.
Problems in Physics of KöMaL, October 2008
