 Problem P. 4148. (March 2009)
Problem P. 4148. (March 2009)
P. 4148. A sample of ideal gas is taken from state A to state B in two different ways. The two states has the same temperature.
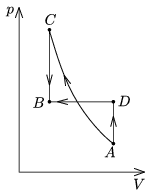
a) First it is compressed adiabatically (AC), and then it is cooled down such that its volume remains constant (CB);
b) First it is heated at constant volume (AD), then it is compressed isobarically (DB).
In which case do we perform less work? At state C or at state D will the temperature of the gas be greater?
(5 pont)
Deadline expired on April 14, 2009.
Statistics:
44 students sent a solution. 5 points: Bartha Zsolt, Földes Imre, Hartstein Máté, Laczkó Zoltán Balázs, Lovas Lia Izabella, Szabó 895 Dávid. 4 points: Egyed Zsombor. 3 points: 16 students. 2 points: 3 students. 1 point: 4 students. 0 point: 14 students.
Problems in Physics of KöMaL, March 2009
