 Problem P. 4212. (December 2009)
Problem P. 4212. (December 2009)
P. 4212. The ratio of the components in a mixture, which consists of two elements, is to be determined. The atomic number of the elements are big, their atomic mass numbers are the same. and the amount of the sample is only 0.8 mg. We know that both elements 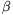 decay when they are bombarded by neutrons. They behave similarly when absorbing neutrons. The half-life of element A is half an hour and the half-life of element B is an hour. Right after the neutron irradiation the
decay when they are bombarded by neutrons. They behave similarly when absorbing neutrons. The half-life of element A is half an hour and the half-life of element B is an hour. Right after the neutron irradiation the 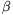 emission is measured. At this time -- subtracting the background -- 400 particles are measured in 10 seconds, and after an hour only 145 particles are measured during 10 seconds.
emission is measured. At this time -- subtracting the background -- 400 particles are measured in 10 seconds, and after an hour only 145 particles are measured during 10 seconds.
Determine the mass of the element A and the mass of the element B in the sample.
(5 pont)
Deadline expired on January 11, 2010.
Sorry, the solution is available only in Hungarian. Google translation
Megoldás. \(\displaystyle m_{A}=0{,}35~\)mg; \(\displaystyle m_{B}=0{,}45~\)mg.
Statistics:
38 students sent a solution. 5 points: Batki Bálint, Benyó Krisztián, Bolgár Dániel, Börcsök Bence, Csopor Dávid, Filep Gábor, Fonai Dániel, Galzó Ákos Ferenc, Hartstein Máté, Janosov Milán, Kovács 444 Áron, Lájer Márton, Lőrincz Dóra, Patartics Bálint, Pázmán Koppány, Szabó 928 Attila, Szöllősi Márton, Tamási Mátyás, Vécsey Máté, Vuchetich Bálint. 4 points: Barta Szilveszter Marcell, Budai Ádám, Farkas Martin, Garaguly Gergő, Iglói Gábor, Jéhn Zoltán, Kungl Ákos Ferenc, Laczkó Zoltán Balázs, Mayer Martin János. 3 points: 5 students. 2 points: 2 students. 1 point: 1 student. 0 point: 1 student.
Problems in Physics of KöMaL, December 2009
