 Problem P. 4740. (May 2015)
Problem P. 4740. (May 2015)
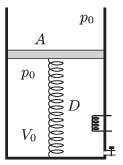
P. 4740. There is a sample of ideal gas in a vertical cylinder, which is closed at its lower base. At the top there is a massless frictionlessly moveable piston. The volume of the sample of ideal gas is \(\displaystyle V_0=8~\rm dm^3\), and its pressure is the same as the ambient air pressure, which is \(\displaystyle p_0=10^5\) Pa. The piston is attached to the bottom of the cylinder with a spring of spring constant \(\displaystyle D=1000\) N/m. The area of the bottom of the cylinder is \(\displaystyle A=2~\rm dm^2\). During a physics experiment the mass of the gas is increased by 50% through a tap on the cylinder, and by heating the absolute temperature of the gas is increased by 60%. Determine the volume and the pressure of the gas after these changes.
(4 pont)
Deadline expired on June 10, 2015.
Statistics:
45 students sent a solution. 4 points: Asztalos Bogdán, Balogh Menyhért, Bartók Imre, Bekes Nándor, Bencsik Bálint, Berta Dénes, Blum Balázs, Boldizsár Bálint, Bugár 123 Dávid, Csathó Botond, Csorba Benjámin, Fehér Balázs, Fekete Balázs Attila, Fekete Panna, Holczer András, Iván Balázs, Jakus Balázs István, Kasza Bence, Körtefái Dóra, Mándoki László, Marosvári Kristóf, Molnár Elizabet, Nagy 555 Botond, Németh Flóra Boróka, Pázmán Előd, Sal Kristóf, Sallai Krisztina, Szántó Benedek, Szentivánszki Soma , Szépfalvi Bálint, Tanner Martin, Varga-Umbrich Eszter, Vastag Balázs, Vida Máté Gergely, Wesniczky Albert, Wiandt Péter, Zöllner András. 3 points: Ardai István Tamás, Horváth Péter. 2 points: 6 students.
Problems in Physics of KöMaL, May 2015
